PARAQUAT



 百草枯
百草枯
Introduction: A herbicide used to control common weeds in a wide range of agricultural and amenity situations.
Common name: Paraquat
Another name: starfire, paraquat ion, Dimethyl viologen, Paraquat dication, Weedol, Spraytop-graze, Dextrone X, etc.
Chemical name: 1,1'-dimethyl-4,4'-bipyridinium
Empirical formula: C12H14Cl2N2
Structural formula:
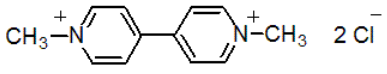
Mol. Weight: 257.2 g/mol
CAS No.: 4685-14-7
Specifications
Leading Paraquat supplier
Paraquat 42% TC
Packing:
BULK PACKING
Liquid: 200L/Drum, 20L/Drum, 10L/Drum ect.
SMALL PACKING
Liquid: 5L/Drum, 1L/Bottle, 500ml/Bottle, 250ml/Bottle, 100ml/Bottle, 50ml/Bottle etc.
Customerized packing label
Paraquat FAO standard
Professional registration
HAZARDS IDENTIFICATION
Hazard statement(s)
H301: Toxic if swallowed.
Precautionary statement(s)
P264: Wash ... thoroughly after handling.
P270: Do not eat, drink or smoke when using this product.
P301+P310: IF SWALLOWED: Immediately call a POISON CENTER/doctor/...
P321: Specific treatment (see ... on this label).
P330: Rinse mouth.
P405: Store locked up.
P501: Dispose of contents/container to ...
Supplemental Hazard Statements: none
MAMMALIAN TOXICOLOGY
Acute toxicity: 1) Acute oral LD50 for mouse: 110 a.i.mg/kg. 2) Acute dermal LD50 for rat: >200 a.i.mg/kg. 3) Inhalation LC50 (4 h) for rat: 0.6 a.i.mg/L. 4) Moderately irritation to eye (rabbits). 5) Slightly irritation to skin (rabbits). 6) Not a skin sensitiser (guinea pigs).
NOEL: (1 y) for dogs is 0.65 mg/kg b.w. daily; (2 y) for rats is 1.7 mg/kg b.w. daily.
Classification:
Toxicity class WHO (a.i.): II (Moderately hazardous)
EC Risk Classification: T+ - Very toxic: R26; T - Toxic: R24/25, R48/25; Xi - Irritant: R36/37/38; N - Dangerous for the environment: R50, R53
ECOTOXICOLOGY
Effect on birds: high toxicity to birds, acute oral LD50 is 35 a.i.mg/kg. Effect on fish: moderate toxicity to fish, acute 96 hour LC50 for Rainbow trout is 19 a.i.mg/L. Effect on aquatic invertebrates: moderate toxicity to aquatic invertebrates, acute 48 hour EC50 for Daphnia magna is 4.4 a.i.mg/L. Effect on algae: high toxicity to algae, acute 72 hour EC50 for Raphidocelis subcapitata is 0.00023 a.i.mg/L. Effect on honeybees: moderate toxicity to honeybees, contact acute 48 hour LD50 is 9.26 a.i.μg/bee, oral acute 48 hour LD50 is >9.06 a.i.μg/bee. Effect on earthworms: low toxicity to earthworms, acute 14 day LC50 for Eisenia foetida is >1000 a.i.mg/kg.
ENVIRONMENTAL FATE
Paraquat's production may result in its release to the environment through various waste streams; its use as a restricted use broad spectrum herbicide will result in its direct release to the environment. If released to air, a vapor pressure of <7.5×10-8 mm Hg at 25 deg C indicates paraquat will exist solely in the particulate phase in the atmosphere. Particulate-phase paraquat will be removed from the atmosphere by wet and dry deposition. Paraquat does not contain chromophores that absorb at wavelengths >290 nm and, therefore, is not expected to be susceptible to direct photolysis by sunlight. If released to soil, paraquat is expected to have no mobility based upon reported Koc values of 15,473-1,000,000. Field studies and soil column leaching studies have demonstrated that paraquat does not leach under environmental conditions. Volatilization from moist soil surfaces is not expected based upon an estimated Henry's Law constant of <3.0×10-14 atm-cu m/mole. Paraquat is not expected to volatilize from dry soil surfaces based upon its vapor pressure. The biodegradation of paraquat is very slow when strongly adsorbed to clay or organic matters in soils because it becomes unavailable. When not in the absorbed-state, paraquat will biodegrade. Some photodegradation may occur on plant leaves or soil surfaces exposed to sunlight. Field dissipation half-lives in soil are reported as 99-4747 days. If released into water, paraquat is expected to adsorb to suspended solids and sediment based upon the reported Koc values. Volatilization from water surfaces is not expected to be an important fate process based upon this compound's estimated Henry's Law constant. Reported BCFs of 0.05-6.9 suggest bioconcentration in aquatic organisms is low. Hydrolysis and direct photolysis in water are not expected based upon laboratory studies.
Usage: Herbicidal properties of the dichloride and bis(methyl sulfate) described by R. C. Brian (Nature (London), 1958, 181, 446) and their properties reviewed by A. Calderbank (Adv. Pest Control Res., 1968, 8, 127). Both salts (though only the former is still sold) were introduced by ICI Plant Protection Division (now Syngenta AG). Herbicidal properties of paraquat dichloride discovered in 1955, and first marketed in 1962. Patents: GB 813531. Manufacturers: Comlets; Crystal; Hesenta; Kuo Ching; Pilarquim; Sanex; Sinon; Sundat; Syngenta; United Phosphorus. Biochemistry: During photosynthesis, superoxide is generated, which damages cell membranes and cytoplasm. Mode of action Non-selective contact herbicide, absorbed by the foliage, with some translocation in the xylem.
Application: Broad-spectrum control of broad-leaved weeds and grasses in fruit orchards (including citrus), plantation crops (bananas, coffee, cocoa palms, coconut palms, oil palms, rubber, etc.), vines, olives, tea, alfalfa, onions, leeks, sugar beet, asparagus, ornamental trees and shrubs, in forestry, etc. Also used for general weed control on non-crop land; as a defoliant for cotton and hops; for destruction of potato haulms; as a desiccant for pineapples, sugar cane, soya beans, and sunflowers; for strawberry runner control; in pasture renovation; and for control of aquatic weeds. For control of annual weeds, applied at 0.4-1.0 kg/ha.







